An introduction to poroide fungi
3: Micromorphology
In the following sections the microscopic characters in polypores are described and discussed. For a more comprehensive overview of these characteristics see Ryvarden (1991).
Hyphae
The basidiocarp consists of hyphae and for a long time no particular attention was paid to them, although there were occasional remarks regarding their thickness and colour and only very rarely was septation mentioned.
Ames (1913) was the first to realize that hyphae were important for an understanding of polypores, but Corner's (1933) descriptions of the hyphal system gave mycologists a new taxonomic tool, his discovery providing a major step towards a more consistent and reliable classification of this group of fungi.
His classification has been somewhat modified in recent years since experience has shown that hyphal analysis is far more complicated than originally assumed.
Generative hyphae
Generative hyphae are the basic units of the structure as they always are present in a basidiocarp and give rise to all other types. Initial stages of the basidiocarp are composed of such hyphae and in some cases no other types of hyphae develop (monomitic hyphal system). In other cases vegetative or secondary hyphae may arise from generative hyphae (see below) to develop di- or trimitic hyphal systems.
The type of septation of generative hyphae is a very important character in the classification of polypores and in difficult cases, such as species with resupinate basidiocarps, this character has to be observed and recorded before a reliable determination can be reached.
The septum between hyphal cells can be either simple (where lacking a clamp connection) or resultant from the formation of a clamp or, rarely, from several clamps.
Septation of generative hyphae is a fundamental characteristic and usually consistent for a given species - it is rather rare to observe species with both multiple clamps and simple septa on the generative hyphae. Some genera such as Albatrellus include species with both single clamps and simple septa. In the Corticiaceae s. lato this phenomenon is more frequent.
It may be difficult to properly interpret septation if the clamp is just below or above the septum and if seen in face view rather than in the side view thus several septa must be observed to reach a reliable conclusion. In simple septate species, the septation at the branching of hyphae may sometimes look very similar to a clamp because the branch may bend back and pass behind the parent hyphae. To be sure, it is best to look for septa in unbranched hyphae.
The inexperienced student may also be misled by adventitious septa. These arise when the protoplasm dries up inside thick‑walled hyphae, which do not collapse during drying. Such dried protoplasm may then appear as a dark line across the hypha, but will normally be much thinner than the hyphal wall, while the true septum will have the same thickness as the rest of the hyphal wall.
If generative hyphae are difficult to find, they may usually be located in the edges of the dissepiments or along the young, actively growing margins of the basidiocarp. In some species the hyphal width may vary quite considerably while in others it is remarkably uniform. Thus, it is important during a microscopic examination to note this and the hyphal diameter.
Branching in some species is quite distinctive, but often difficult for a beginner to grasp properly. In species like Phaeolus schweinitzii hyphae branch off in almost right angles, but this is unusual and normally there will be an acute angle between the main stem and the new side‑branch.
In the context of many species with resupinate basidiocarps there will exist H‑connections, these resembling a small hyphal bridge between two more or less parallel hyphae. These are of no taxonomic significance. The wall thickness of generative hyphae may vary considerably although they are mostly thin‑walled. In some species however, a considerable thickening may be observed and in such cases non‑septate segments may easily be taken as skeletal
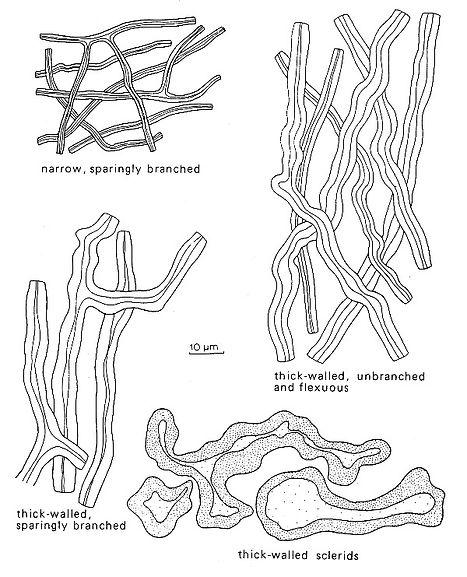
Fig. 5. Types of skeletal hyphae and sclerids. Del. R. Gilbertson.
The wall thickness of generative hyphae may vary considerably although they are mostly thin‑walled.
In some species however, a considerable thickening may be observed and in such cases non‑septate segments may easily be taken as skeletal hyphae. Such hyphae are most common in the context of monomitic species, such as Ischnoderma resinosum and are often called sclerified generative hyphae - it is thus important to examine at least some hyphae over a considerable length to exclude the occurrence of clamps or simple septa before they are defined as skeletal hyphae instead of sclerified generative hyphae.
Also, since generative hyphae give rise to all other types, it is not unusual to see segments of the same hypha with an abrupt transition between generative and skeletal elements, separated by a clamp.
Gloeoplerous hyphae
In some polypores there may be wide, thin‑walled hyphae with a refractive content these distinctly different from the normally hyaline generative hyphae. Such hyphae are called gloeoplerous hyphae. They are often yellowish, with homogeneous or grainy contents. In some cases the content darken in Melzer’s reagent or stain brightly with phloxine. The type of septation at each end of these hyphae is often very difficult to observe, but in some cases large, distorted clamps are present. The occurrence of gloeoplerous hyphae is rather scattered in Polyporaceae s. lato, and they have taxonomic significance only at species level.
Vegetative hyphae
Vegetative hyphae have, by definition, no septa. They arise from a septum or clamp and may (for instance with binding hyphae) be of restricted growth, or in the case of skeletal hyphae be of unlimited growth. In rare cases they may be intercalary i.e. a skeletal hypha may arise from a septum and may end abruptly via a septum developing further as a generative thin‑walled hypha.
In trimitic species, it is usually easy to distinguish skeletal hyphae from binding hyphae. However, in many species this is not the case as there are transitions from dichotomously branched skeletal hyphae to more arboriform types which come very close to binding hyphae of the type seen in Polyporus s.str. In such cases the different aspects of the vegetative hyphae are described in detail and it is a matter of opinion whether such hyphae should be classified as skeletal or binding hyphae proper.
Skeletal hyphae
A skeletal hypha is normally unbranched and thick‑walled to solid. In some species however, it is possible to find sparingly branched solid hyphae without septa, which in this manual will be called skeletal hyphae. Normally a skeletal hypha will have an even diameter along most of its length while a genuine binding hypha normally has tapering side branches arising from a main stem.
Arboriform skeletal hyphae occur in the genus Ganoderma. They are unbranched up to 200 µm and then form tree‑like branches. If loose segments of such hyphae are observed separately, they may easily be taken for arboriform binding hyphae. In some species it is necessary to examine long segments of suspected skeletal hyphae to be sure that no clamps or simple septa occur and that sclerified generative hyphae are not misinterpreted. Only experience will help here, because such hyphae can be very similar to skeletal hyphae proper.
Under a phase contrast microscope skeletal hyphae are light‑refractive because of the thick walls. At the apex they will be thin‑walled, often becoming so rather abruptly from the normally thick‑walled parts. Some adventitious septa may occur at the apices as they are more densely filled with protoplasm than the older parts.
Skeletal hyphae should be mounted in Melzer’s reagent, because in some species they react and change colour to strongly reddish brown (dextrinoid reaction) or greyish‑bluish (amyloid reaction). As Melzer’s reagent itself is pale yellowish, it is inevitable that hyphae mounted in it will be also be slightly coloured when viewed in this reagent. Such a weak colouration should not be confused with a dextrinoid reaction.
When mounted in 3‑5% KOH the skeletal hyphae of some species, such as Cinereomyces lindbladii, become gelatinized or strongly swollen with blurred outlines. They may even break down completely in KOH, as is the case in Piptoporus soloniensis. A change of mounting medium is then necessary to ascertain the true nature of the vegetative hyphae. The use of KOH has advantages for those working with a phase contrast microscope, but both spores and hyphae have a tendency to swell slightly and give measurements about 1 µm larger than those reported from measurements in Melzer's reagent, Cotton blue and other media.
Binding hyphae
Binding hyphae are branched, very thick‑walled or even solid and non‑septate, often with a more randomly oriented growth than skeletal hyphae. In trimitic genera such as Fomes and Trametes, they are intricately intertwined between the more radially (in the context) or vertically (in the trama) oriented skeletal hyphae.
Such binding hyphae are shortly branched with tortuous side branches - it is difficult to observe their origin. They are normally thinner and far more sinuous than skeletal hyphae. In some cases it is believed that they originate as side branches of skeletal hyphae.
Arboriform binding hyphae of the Bovista‑type [very similar to the type of binding hyphae observed in some gasteromycetes (e.g. Bovista)] are present in Polyporus and Dichomitus. They have a tree‑like branching and distinctly tapering side branches. Among the polypores, skeletal hyphae are much more widespread than binding hyphae.
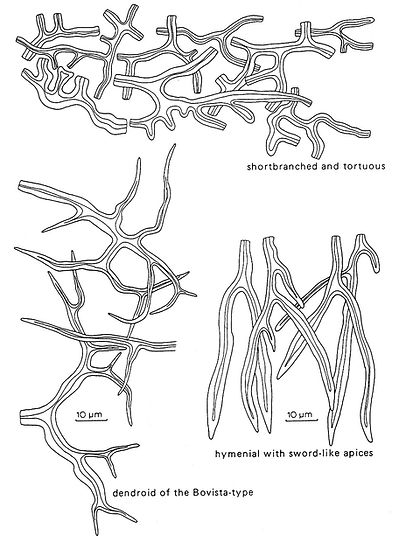
Fig. 6. Types of binding hyphae. Del. R. Gilbertson.
Hyphal construction of the pileal cover
In species with a distinct pileal cover, it may often be useful to check the hyphal system in the tomentum.
In most trimitic species the tomentum will consist of skeletal hyphae mixed with some scattered generative hyphae. In monomitic species, the tomentum is composed of generative hyphae, often with a considerable thickening of the walls and
wider than those of the trama.
In Polyporaceae and Hymenochaetaceae a cuticle or crust will normally be composed of densely agglutinated hyphae without diagnostic value.
In numerous species in the Ganodermataceae a palisade of hyphal ends may be seen, the shape and size of which can be important. The hyphae arise from clamped generative hyphae, but the clamps generally disappear as the walls increase in thickness to 3 or 4 µm. A few species such as Coltricia perennis have distinctively branched, antler like elements on the pileus surface.
The hyphal construction of the context
The hyphal construction of the context is important as in many species it differs from that of the trama, and is thus of diagnostic value.
It may also change considerably during different stages of development. For example, the context of young, immature basidiocarps of Polyporus squamosus consists mainly of generative hyphae with conspicuous clamps whereas in mature specimens it is composed mainly of binding hyphae with generative hyphae difficult to discern.
In species with vegetative hyphae the context will eventually be composed almost entirely of skeletal hyphae, and will remain so in the dimitic species. However, in the trimitic species binding hyphae will develop as the specimen matures.
Thus, a fragment from the basal parts of the context will normally include all types of hyphae present in the context.
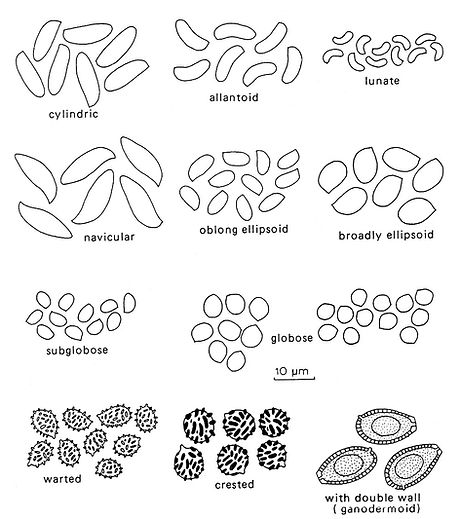
Fig. 7. Basidiospore shape and ornamentation. Del. R. Gilbertson.
Hyphal construction of the trama
The trama is, in many species, of a different construction than the context, which will often be apparent to the naked eye as it is a different colour from the context.
As the trama is developed vertically the hyphae are normally more or less parallel, but in some species the vegetative hyphae may be intricately intertwined and difficult to separate. Thus, it is important to take a section along the trama to ascertain the general structure.
In some genera such as Phellinus there are striking differences in tramal construction, from one species to another. Vegetative hyphae are normally confined to the central trama being surrounded by generative hyphae to form a subhymenium from which the basidia are developed.
Besides generative hyphae and vegetative hyphae (mostly skeletal hyphae), gloeoplerous hyphae may occur. As stated under the section on cystidia (below) skeletal hyphae may project or bend into the hymenium and will then be called cystidia. Setal hyphae in some species of Phellinus and Inonotus may occur in the same way and are rather conspicuous.
Structure of the dissepiments
In most species the dissepiments are composed of sterile hyphal ends, either generative alone or mixed with skeletal hyphae. These hyphal ends may be encrusted, as in Skeletocutis and could then be easily interpreted as some sort of cystidia. However such an encrustation is normally irregular with scattered crystals, as in Schizopora, or dense as in Skeletocutis species on most hyphae. These will then be termed as encrusted hyphae rather than cystidia.
In a few species such as Datronia mollis and D. stereoides the dissepiments contain finely branched dendrohyphidia (Fig.8) often covered with fine crystals that obscure their true nature. Careful observation is necessary to reveal their presence.
Basidia
Basidia are the hymenial cells in which fusion of nuclei or karyogamy, and meiosis occur, and which bear the basidiospores on terminal projections called sterigmata.
Like other hymenomycetes, polypores have a continuous development of basidia over periods varying from several days to many years. Consequently basidia in all stages of development may be found in any actively sporulating basidiocarp.
Most polypores have 4‑spored (tetrasterigmatic) basidia which are more or less clavate and hyaline. Thus, their shape is of little diagnostic value in this group. However, their size varies considerably from species to species and may be an important taxonomic character. If clamped generative hyphae are present in the context and trama, the basidia will have a basal clamp.
In order to determine the true length of basidia from the basal septum to the apex it is necessary to crush out some very thin sections to separate individual basidia. This is somewhat difficult to do and provides a good test for the skill of the polyporologist.
Basidiospores
Basidiospores are accepted as important for classification of all basidiomycetes, thus for the polypores. Specimens collected when active sporulation is not in progress may have few or apparently no spores at all and this is one of the most difficult problems in the identification of polypores.
The ability to cut good thin sections and to find basidiospores is a skill that anyone interested in polypores must have. Many species may be reliably determined when sterile, but for others examination of spores is necessary for a determination. This is especially the case for species with resupinate basidiocarps.
Spore size
Measurements are given as a range of variation based on 10 measurements or more for each species. It is important to note that a few spores may be larger or smaller than the range given for a particular species. Larger spores can be a result of the development of basidia producing less than 4 spores, whilst smaller spores are often simply immature.
A spore print is desirable since spores are never released in nature before they are mature. It should also be noted that spores measured in KOH will often be slightly larger than in Melzer’s reagent! For ornamented spores measurements given in this book include the spines or aculei.
Spore shape
The shape of basidiospores is important and Fig. 7 shows some of the more common types of spores and their designation. Spores tend to become longer and narrower as they mature and this may account for a degree of variation.
Spore ornamentation and wall thickness
Basidiospores of most polypores are smooth and thin‑walled, but in a few genera they are ornamented with small warts, spines or longitudinal striae.
In the Ganodermataceae, spores are very distinctive with a thick yellowish to pale brown endospore, on which there is a regular pattern of warts or protuberances. Covering the endosporic protuberances is a hyaline exospore which is invisible in microscopic preparations. In SEM pictures it is apparent as a thin "cover" around the spores.
The ornamentation on some spores may be difficult to observe in KOH, and sections should be mounted in either Melzer's or another medium before a spore is stated to be smooth. Especially for species with amyloid spores, it is necessary to observe them very accurately as many spores with an amyloid reaction have very minute ornamentation. Observation with oil immersion or a scanning electron microscope may be essential to discern the ornamentation. Basidiospores with thickened walls are easiest to observe in cotton blue, Melzer's or phloxine. Under a phase contrast microscope spores mounted in Cotton Blue will appear almost red if they are thick‑walled. In KOH it may be difficult to observe the thickness of the wall properly.
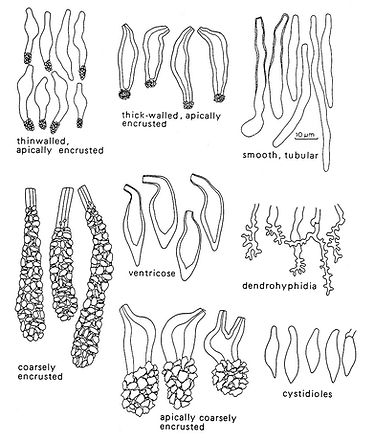
Fig. 8. Types of cystidia and other sterile hymenial organs. Del. R. Gilbertson.
Cystidia and other sterile hymenial organs
In many polypores, as well as in other basidiomycetes, sterile organs occur among the basidia and are of importance in delimiting genera and identifying specimens. They have different designations according to where they arise and their morphology.
Cystidia
Cystidia may be divided into two groups according to where they arise. Hymenial cystidia arise in the subhymenium and are usually of the same size or slightly larger than basidia. They may be thick‑ to thin‑walled, some are smooth while others have a crown of crystals or may even be encrusted for a considerable length. Their shape may, as the figures show, vary from clavate to ventricose. They will mostly be prominent and easy to observe, but may also be rare. In such cases the species is keyed out at least twice. In a very few species (as in the genus Amylocystis) the cystidia react in Melzer’s reagent.
Tramal cystidia arise, as the name indicates, in the trama, and may (or may not) bend into the hymenium, often protruding far beyond it. Usually they are the outer ends of skeletal hyphae with a widened and often strongly encrusted apex. Sometimes such cystidia also occur in the pore mouths or may be found deeply embedded in the trama. They are often called skeletocystidia.
Gloeocystidia are smooth and thin‑walled cystidia, often tubular and slightly sinuous or constricted with grainy to oily, refractive content. They are generally conspicuous in Melzer's reagent and usually stain brightly in phloxine.
Gloeocystidia are analogous to gloeoplerous hyphae and in some cases there are transitions between them. However, if such structures are more or less confined to the hymenium, they are called gloeocystidia. They will be far more refractive and normally larger than sterile hyphal ends which may occur in the hymenium. Gloeocystidia are relatively uncommon in polypores.
Cystidioles
Cystidioles are smooth, bottle‑shaped or clavate, often pointed organs of the same size as the basidia. They occur amongst the basidia, in the hymenium, in some species in large numbers but are only diagnostic at the specific level. They may be distinguished from immature basidia by their pointed and tapering shape. Immature basidia are slightly widened towards the apex and have a rounded top. It may be that cystidioles represent aborted basidia where meiosis has failed.
Dendrohyphidia
Dendrohyphidia are branched organs that may occur in the hymenium and edges of the dissepiments in a few genera of polypores (e.g. Datronia). Arising from a septum, their degree of branching may vary quite considerably. The outer apices are mostly very thin‑walled and collapse rapidly on drying. In many cases they are strongly encrusted with tiny crystals. Thus, they can be easily overlooked unless specifically looked for. Their function is unknown although they may play a role in excretion of metabolites (like cystidia).
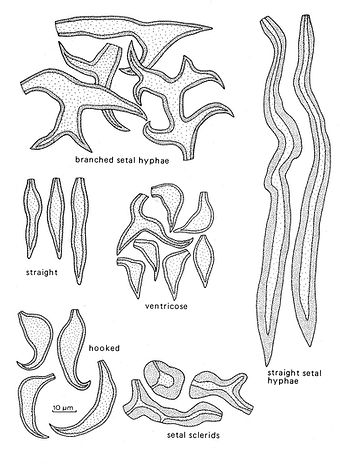
Fig. 9. Types of setae and setal hyphae. Del. R. Gilbertson.
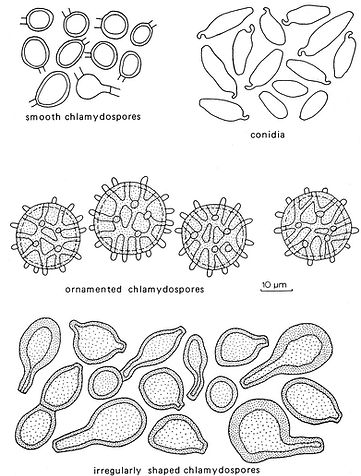
Fig. 10. Types of asexual spores. Del. R. Gilbertson.
Setae and setal hyphae
Setae and setal hyphae are confined to the Hymenochaetaceae, are diagnostic for the family, and play an important taxonomic role at the specific level. They are dark brown, thick‑walled and prominent when they occur.
Setae
These are of two types, hymenial and tramal. The former occur in the hymenium, arise at the same level as the basidia, and usually project beyond them, in some species very conspicuously so. They may then be observed with a good hand lens. In most cases they are simple with a sharp and tapering apex. The apex may be straight or distinctly bent or hooked.
Hymenial setae arise terminally from hyphae, but may also be lateral. In the latter case they will be extended laterally from the base on both sides and may often be somewhat swollen at the base.
Setae are either subulate, i.e. broadest at the base and tapering evenly towards the top or ventricose, i.e. with a swollen part above the base and then more abruptly constricted at the top. In most species the shape of the setae is fairly constant, whilst in others there may be a mixture of subulate and ventricose ones and a clear distinction may then be difficult to draw.
Tramal setae are embedded in the trama and run parallel to the tube walls or may project obliquely into the hymenium. They may be of considerable length and could be called giant setae. It is easy to confuse them with setal hyphae (see below), but true tramal setae are distinctly conical and are widest in the middle part and taper slightly towards the apex. It must be admitted, that in some cases it is difficult to separate them from short setal hyphae.
Setal hyphae
When present these organs are conspicuous and may be up to 400 µm long and 25 µm wide. They are often straight and run parallel to the tubes, but in some species the apex is bent into and beyond the hymenium and may then be seen with a good hand lens. Setal hyphae are of equal width tapering only at the apex and are thus, different from the tramal setae. They may also be present in the context, especially in marginal tissue, and in mycelial felts in decayed wood. Branched setal hyphae occur on the pileal surface of a few species of Inonotus.
Asexual spores
Asexual spores are rare among the polypores, but rather important in those few species where they occur.
Conidia
Conidia are rare in the polypores and only in a few cases are they diagnostic. They develop from conidiophores or undifferentiated conidiogenous hyphae, often in the margin or in the context, but their occurrence is normally so irregular that their presence or absence is not used in the keys.
Chlamydospores
Chlamydospores occur in the basidiocarps of a few polypores and are an important character for identification of those species. Chlamydospores differ from conidia in being thick‑walled and are often intercalary and rarely terminal. They function as a resistant resting stage in the life cycle. In Europe and North America Inonotus rickii has basidiocarps that almost always differentiate into a mass of chlamydospores rather than forrming a poroid basidiocarp and this is also known from a few places in Europe.
Hyphal Pegs
Hyphal pegs occur in quite a number of polypores and can best be seen in sections cut across the tubes. These structures appear as either papillate or columnar projections from the hymenium or may represent ridges that run longitudinally and appear as pegs in cross sections. They are composed of intertwined vegetative hyphae and may be readily recognized by the absence of a hymenium on their surface.